| | posted by Suzie Chhouk Learn about the history of the word 'dynamite'. What makes dynamite so spectacular? Nitroglycerin. |
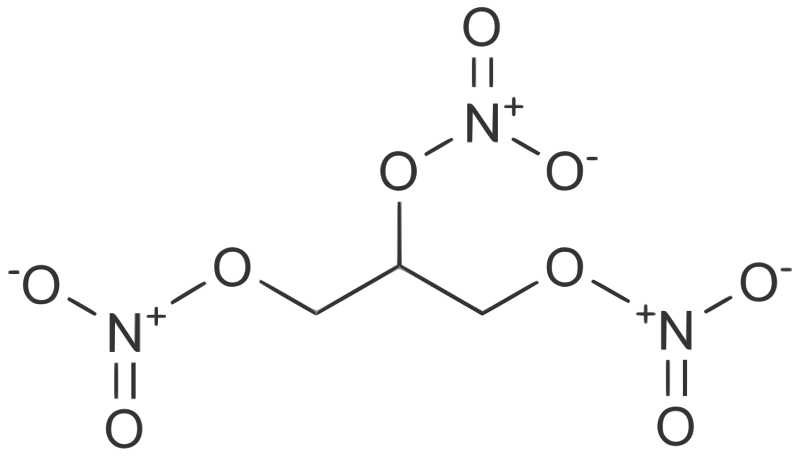
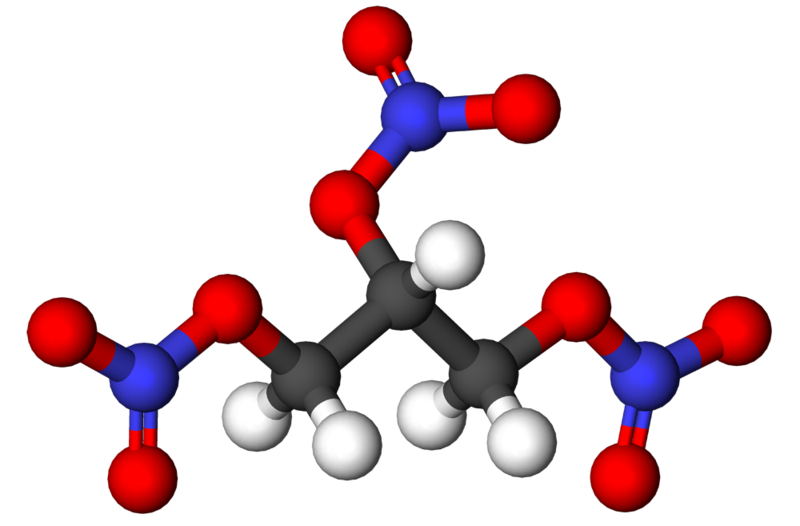
Chemists spell nitroglycerin as C3H5N3O9. 'Glycerin' refers to a three-carbon chain, and 'nitro' refers to the three NO3 groups, one attached to each carbon. Its chemical structure is shown in the figures below.
Properties of nitroglycerin:
Molar mass: 227.0865 g/mol
Appearance (at room temperature): colorless liquid
Density: 1.6 g/mL (at 288 K)
Melting point: 287 K (14 degrees Celsius, 57 degrees Fahrenheit)
Boiling point: 323 K (50 degrees Celsius, 122 degrees Fahrenheit)
Solubility: low in water; high in acetone, ether, benzene, alcohol
Molar mass: 227.0865 g/mol
Appearance (at room temperature): colorless liquid
Density: 1.6 g/mL (at 288 K)
Melting point: 287 K (14 degrees Celsius, 57 degrees Fahrenheit)
Boiling point: 323 K (50 degrees Celsius, 122 degrees Fahrenheit)
Solubility: low in water; high in acetone, ether, benzene, alcohol